Aneuploidies QF-PCR
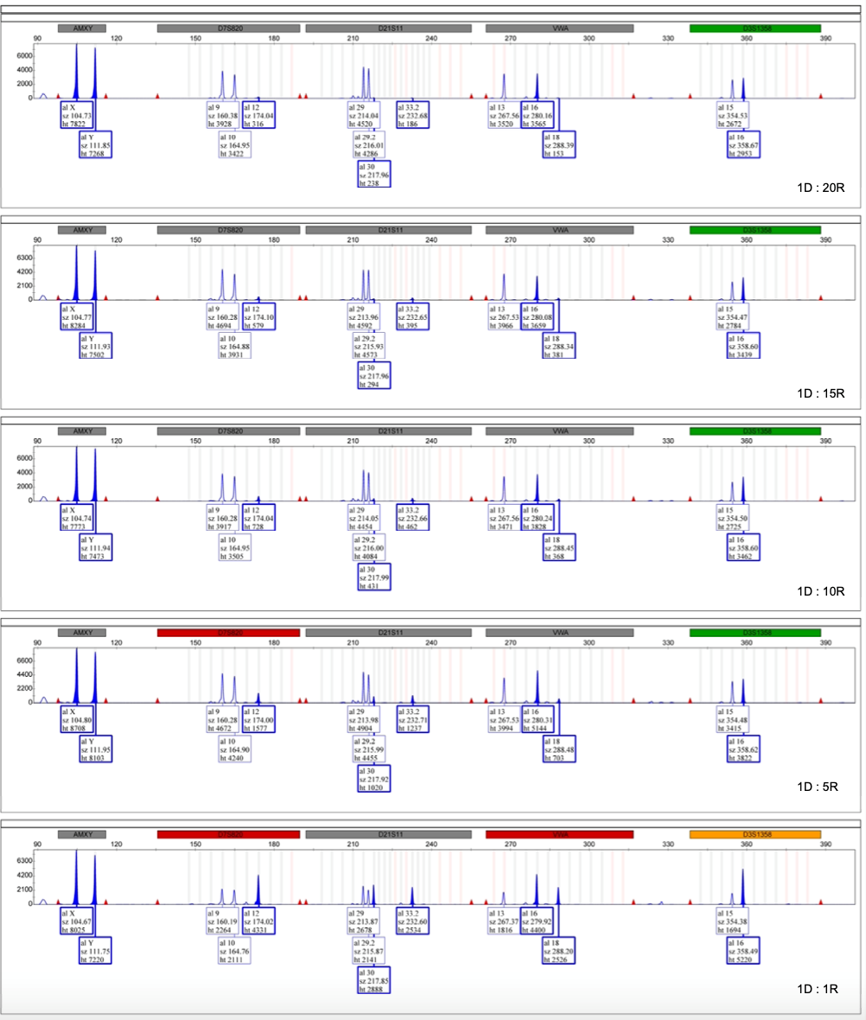
Chromosomal aneuploidies are the most common class of chromosomal aberrations and the main cause of live-born chromosomal disorders. QF-PCR is the current method of choice for rapid and accurate detection of the most prevalent chromosomal aneuploidies, such as trisomy of chromosomes 21, 18, 13, X, and Y, and Turner syndrome (45, X). Genetek Biopharma has developed a family of QF-PCR kits based on analyzing the inheritance of short tandem repeat (STR) markers in fetal samples. To meet the diverse needs of medical genetics laboratories aiming for precision, GT’s line of QF-PCR Aneuploidies detection offers kits with 26–32 markers.
HapScreen Kit
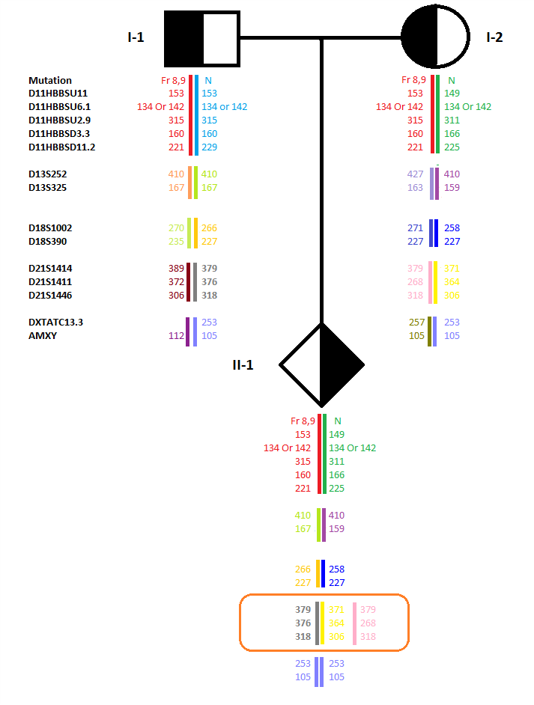
There are numerous risk factors making the procedure of prenatal diagnosis critically prone to errors. Every year, inevitable events such as sample mix-up, maternal cell contamination, non-paternity, possible disomies, new mutations, crossing over, etc., lead to lawsuits regarding the consequent misdiagnoses. To provide a reliable solution to such challenges, Genetek Biopharma has developed an innovative approach to overcome most causes of diagnostic errors using a single multiplex QF-PCR. Each kit contains short tandem repeat (STR) markers linked to a specific gene and STR markers used for QF-PCR to detect aneuploidies of chromosomes 21, 18, 13, X, and Y. GT HapScreen® Kits have been developed to detect various genetic diseases. The main features of GT HapScreen® Kits can be summarized as follows: Tracking disease gene segregation Confirming direct mutation detection results Ruling in/out maternal cell contamination Ruling in/out sample mix-up Verifying sample authenticity Confirming paternity Checking for possible chromosomal aneuploidy.
Male Infertility
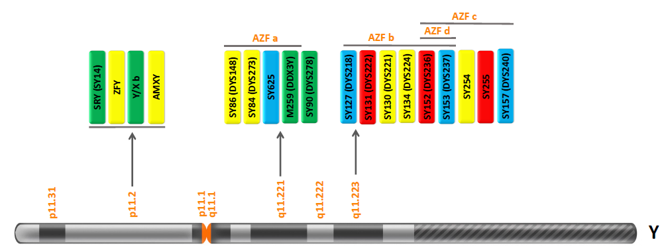
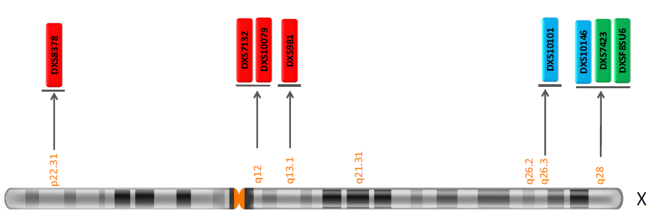
Infertility is a critical problem for most couples. It affects 15-20% of couples of reproductive age worldwide. Male infertility contributes to about 20-30% of such cases, out of which Y chromosome microdeletions cause 5-10% being the most common cause of infertility in men. Deletions in a specific region on the human Y chromosome are responsible for azoospermia named Azoospermia Factor or AZF. These are subdivided into AZFa, AZFb, AZFc, and AZFd. Genetek has developed two types of kits to detect Y chromosome microdeletions: Y chromosome aneuploidy or Klinefelter Syndrome (47, XXY).
Repeat Expansion
Some human genetic diseases are caused by the expansion of repeat units either within a gene or very close to the gene. These expansions interrupt proper protein synthesis or normal protein function. Among these disorders are Fragile X Syndrome, Friedreich Ataxia, Myotonic Dystrophy Type 1, Huntington Disease, etc. The repeat units and expansion differ in each case. The determination of repeat expansion has diagnostic values as well as prenatal diagnosis. The number of repeats can guide the physicians to decide if the expansion is at normal, premutation, intermedia, or full mutation state. Genetek Biopharma has developed several kits for the determination of repeat expansion disorders.
Carrier Screening
Carrier screening has different meanings. One is determining if a person is a carrier of a disease or not. This part has become more important by the implementation of Next Generation Sequencing (NGS) as more couples are requesting to know if both of them carry autosomal recessive genes or if any of them carry an autosomal dominant gene that is present in their offspring.
However, another more important aspect of carrier screening is its role in public health. Some countries have developed carrier screening programs for some diseases such as hemoglobinopathies (e.g., thalassemia, Sickle Cell Disease, etc.), Cystic Fibrosis, Spinal Muscular Atrophy (SMA), etc.
Genetek Biopharma’s carrier screening line of products offers reliable solutions for this purpose.
Chimerism
Bone marrow transplantation has become a routine practice for leukemias, hemoglobinopathies, etc. for few decades. It is essential, for the physicians to assess the success of transplantation and to determine the percentage of donor to recipient cells in the recipient body. Therefore, chimerism is an important issue in transplantation follow up.
Genetek Biopharma has developed an STR-based multiplex PCR kit for chimerism detection and percentage determination.
PGD
Preimplantation Genetic Diagnosis (PGD) has proven to be an extremely useful procedure to ensure that a couple can have a healthy child without worrying about fetal health and therapeutic abortion. In some cases, like thalassemia one can offer PGD to have a non-thalassemic child with the right HLA typing.
Genetek has developed several kits to help those centers providing PGD services. Additionally, as Ezy PGD kits provide all requirements for performing a quick and accurate PGD procedure, they can also benefit laboratories that are setting up their PGD unit for the first time.
Mutation Detection
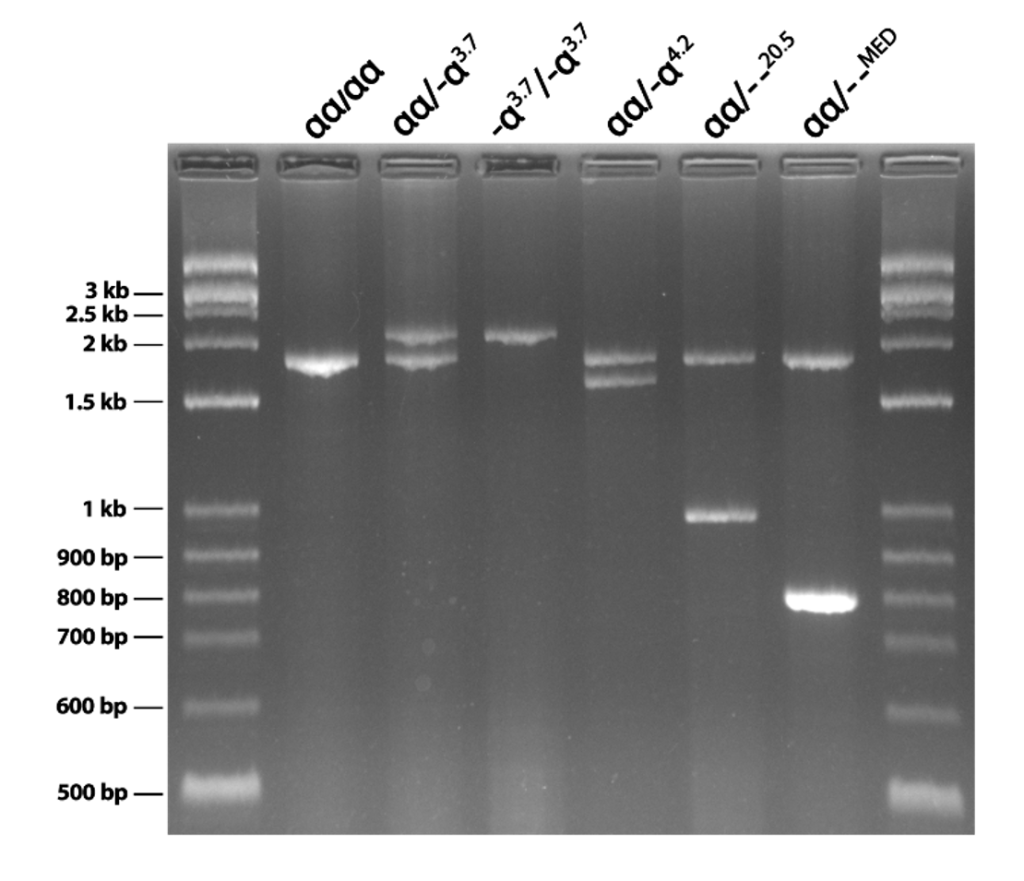
Mutations are one of the main causes of human genetic disorders. Mutations have many forms, from substitutions, deletions, repeat expansions, insertions, methylations, inversions, etc. Rapid and accurate mutation detection is an essential part of medical genetics laboratories.
Genetek Biopharma has been developing kits for rapid and accurate mutation detections. Some of these kits are fluorescent based and others are agarose gel based.