BIOPHARMA
Genetek Product Category
Genetek is an innovative company producing innovative and high quality products for diagnosing human genetics diseases, PGD, infertility in men, human identification and forensic genetics, animal genetics, etc.

Diagnostic Kits
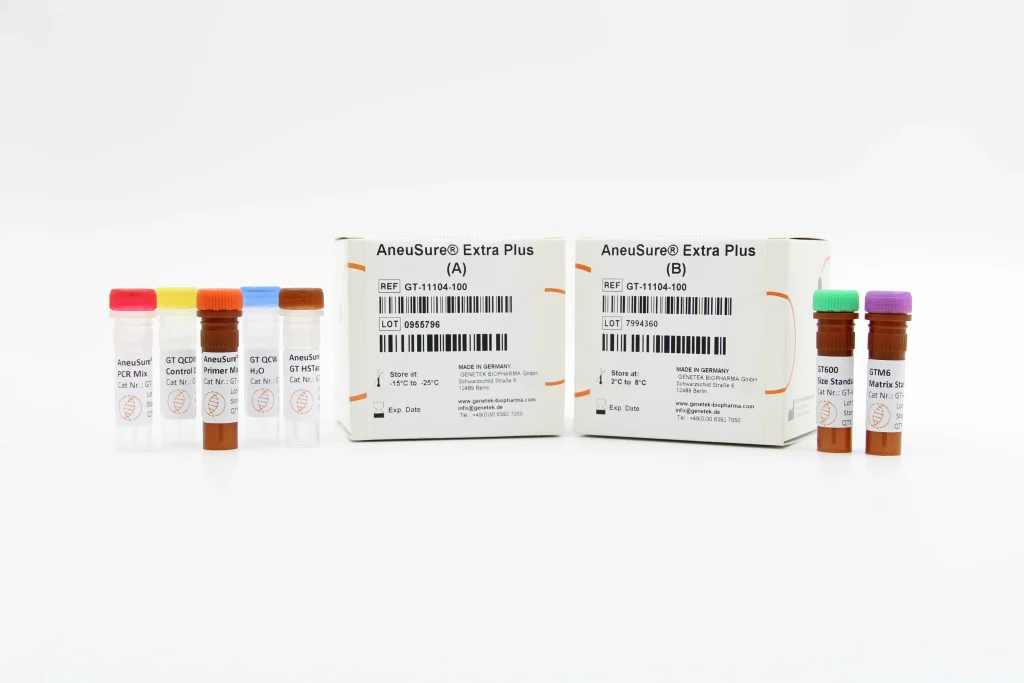
Genetek has kits for diagnosis of human genetic disorders, chromosomal aneuploidies, male infertility, repeat expansion, PGD, etc.
Human Identification
Genetek has kits for human identification, human profiling, and forensic genetics. We have kits for autosomal, X, and Y chromosomes profiling.

Animal Identification
Genetek has kits for animal profiling and identification. Our horse profiling kits have many advantages to other kits with unique properties and ease of use

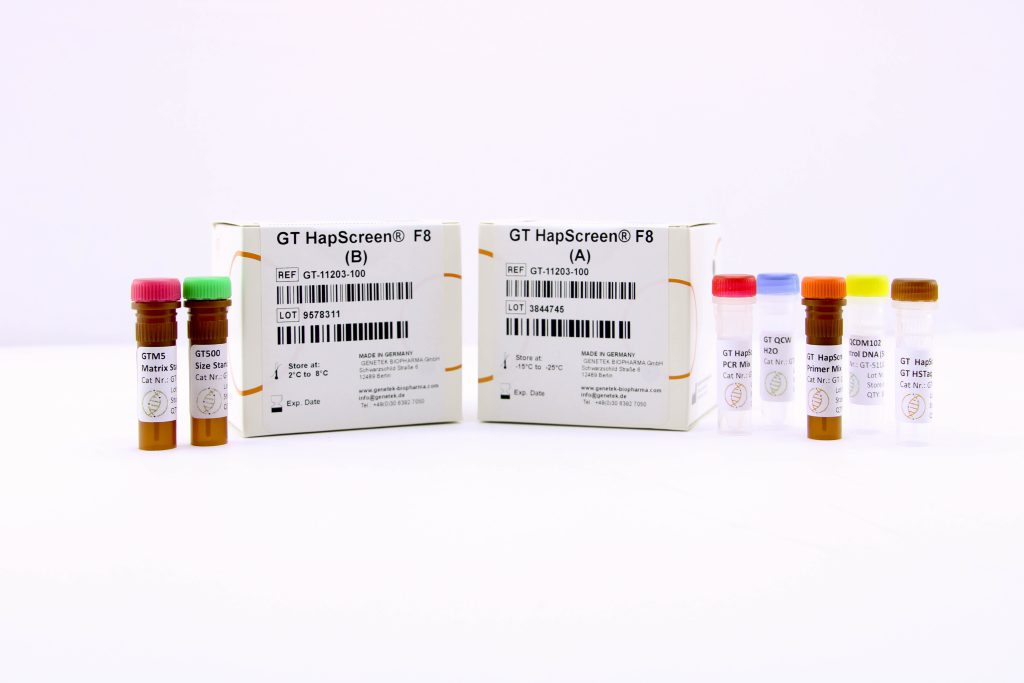
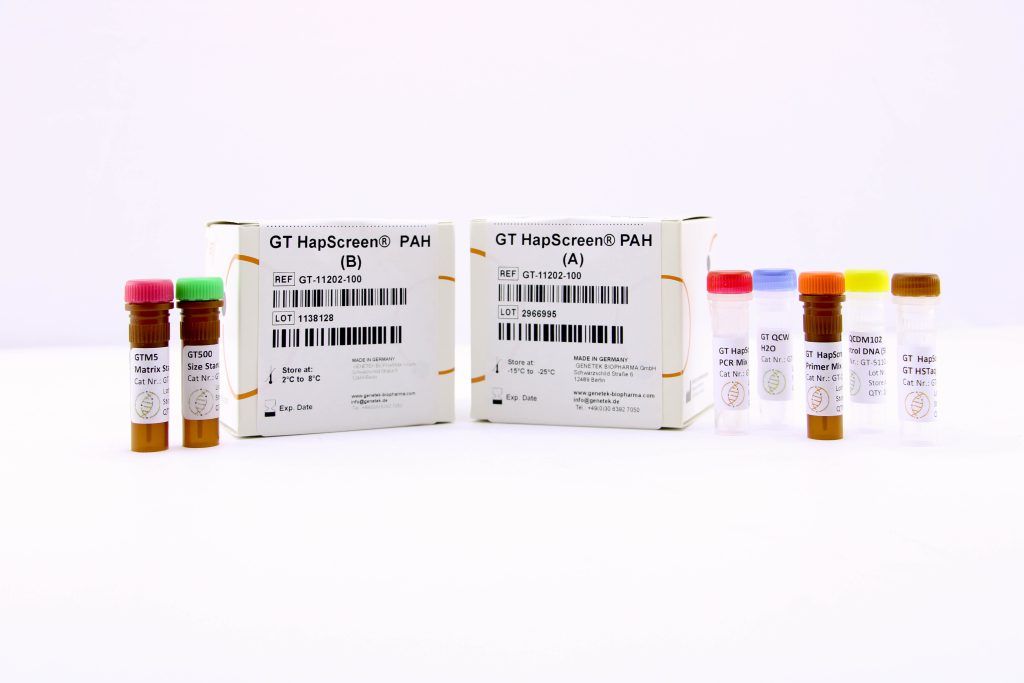
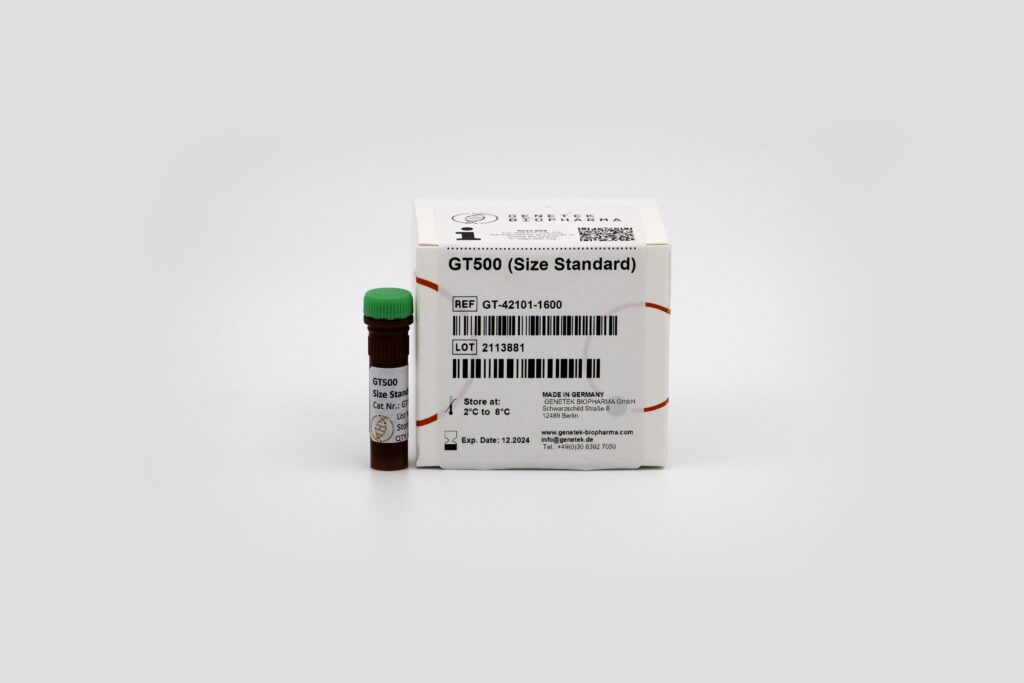
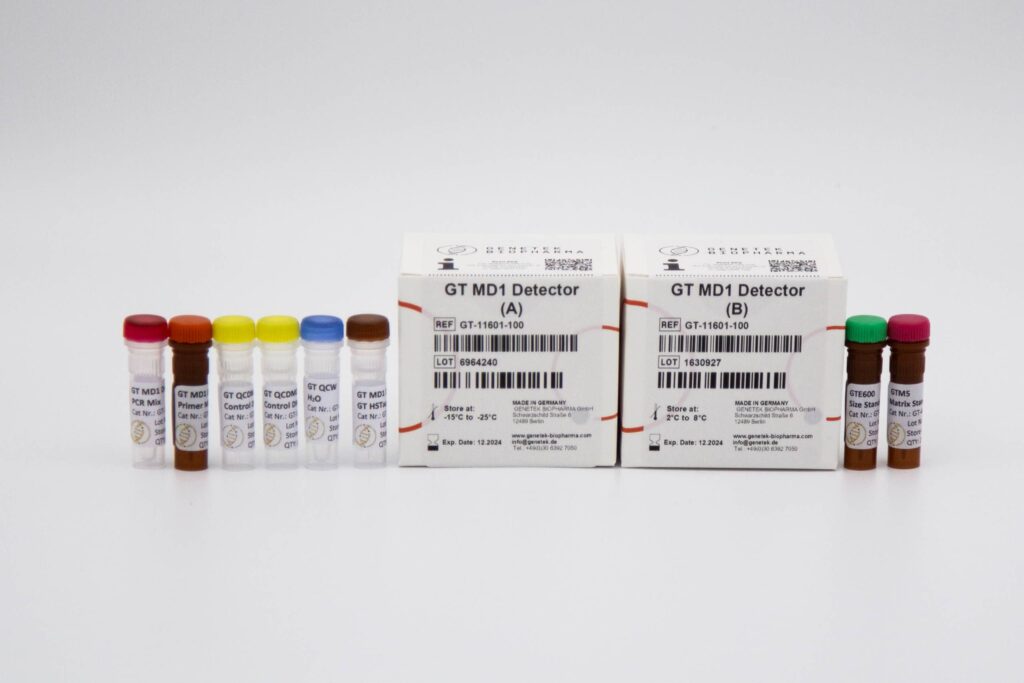
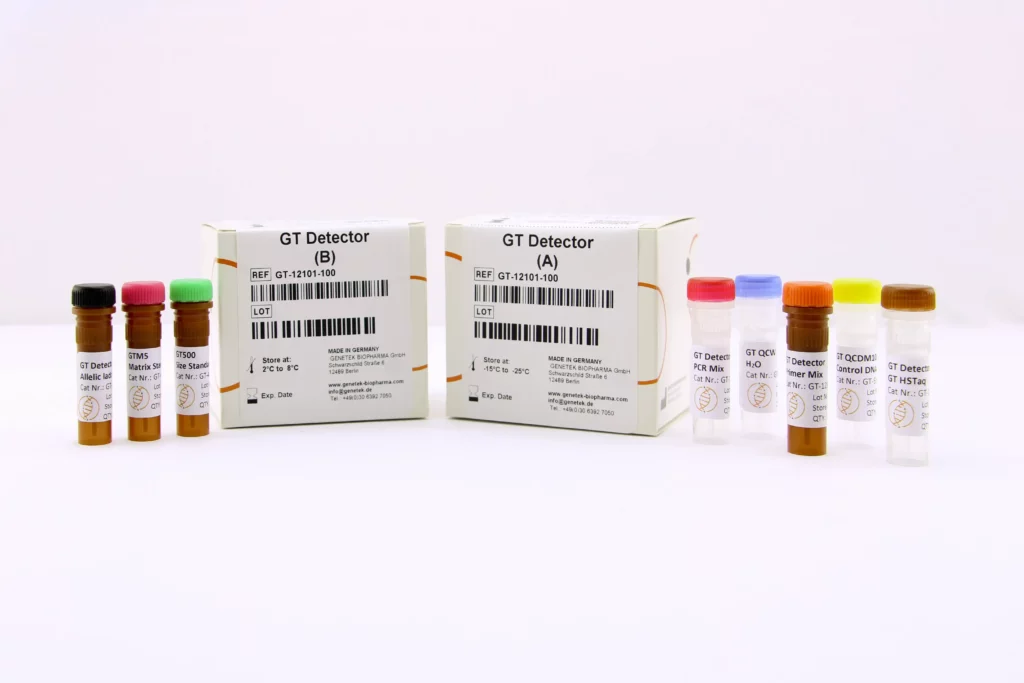
Reagents & Kit Accessories
Our wide range of kits accessories are directly related to our kits
The kit accessories as products which enable the user to have extra hand in performing tests and research
Our Products
Genetek has developed many kits for human disease, infertility, human identification, animal identification, etc.
Research & Collaboration
GENETEK Biopharma GmbH appreciates collaborations to work with academic scientific teams, universities and industrial partners to further novelty for a better posterity.
We are open to partnerships with institutes and/or organizations to facilitate external validation of our in-vitro genetic diagnostic kits, human identification and animal identification kits.
0
research and development
Support
Genetek has many supporting documents and products (including user manuals, presentations, audio and video, posters, etc.) to support our customers. We also have a dedicated team to support, to receive queries, provide help and support to users of our product and customers to have full benefits of our innovative products. Our supports also include user manuals, instrument set-up, quick protocols, charts, presentations, etc. to help you to have the best benefit from Genetek products.
Product support
Do you have trouble in finding the right product? Please get in touch by sending us an email to: info@genetek-biopharma.com
Documents
Although Genetek products come with the relevant documents and supporting materials, however, we welcome any request for a specific document that may not be available or need to send you by email. Please see the document section and if you could not find the right document, contact us at:
Procedure
If you need procedure for a specific product or need a specific protocols, please send us an email:info@genetek-biopharma.com
Blog & News
Follow our Blog for new products, events, news and more
The Power of Horse Genetic Profiling
How GT Equine's STR Marker-Based Kit is Transforming the Equine Industry Understanding STR markers and